AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Helium electron configuration12/16/2023 
It has seven valence electrons.Ĭheck the related article “Is chlorine a metal?”. How many valence electrons does chlorine have?Ĭhlorine has the atomic number 17. As a result, it contains two valence electrons. It has an electrical configuration of 2,8,2. In that sense, a helium atom is described as a molecule as well. Physicists, on the other hand, frequently use the term “molecule” to refer to the smallest particle of gas, regardless of how many atoms or what type of bonds that particle contains. Helium is classified as a noble gas due to its complete outer shell. In addition, Vander Der Waal’s forces are physical bonds, not chemical bonds. A molecule is defined as a group of two or more atoms bound to one another. Helium is not a molecule, but rather a collection of atoms held together by Van Der Waals interatomic forces in fact, only London forces hold helium together. Helium is the only element that cannot be solidified by sufficient cooling at normal atmospheric pressure it requires a pressure of 25 atmospheres at a temperature of 1 K (272 ☌ or 458 ☏) to be applied to convert it to its solid-state. 
Helium has lower boiling and freezing points than any other known substance. Helium, the second lightest element (only hydrogen is lighter), is a colorless, odorless, and tasteless gas that solidifies at 268.9 ☌ (452 ☏). Therefore, there are various non-equivalent definitions of atomic radius.Helium (He) is a chemical element and inert gas in Periodic Group 18 ( noble gases). However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. :max_bytes(150000):strip_icc()/heliumatom-58b602983df78cdcd83d95c8.jpg)
The atomic radius of Helium atom is 28pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Helium are 3 4. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Helium is a chemical element with atomic number 2 which means there are 2 protons in its nucleus. 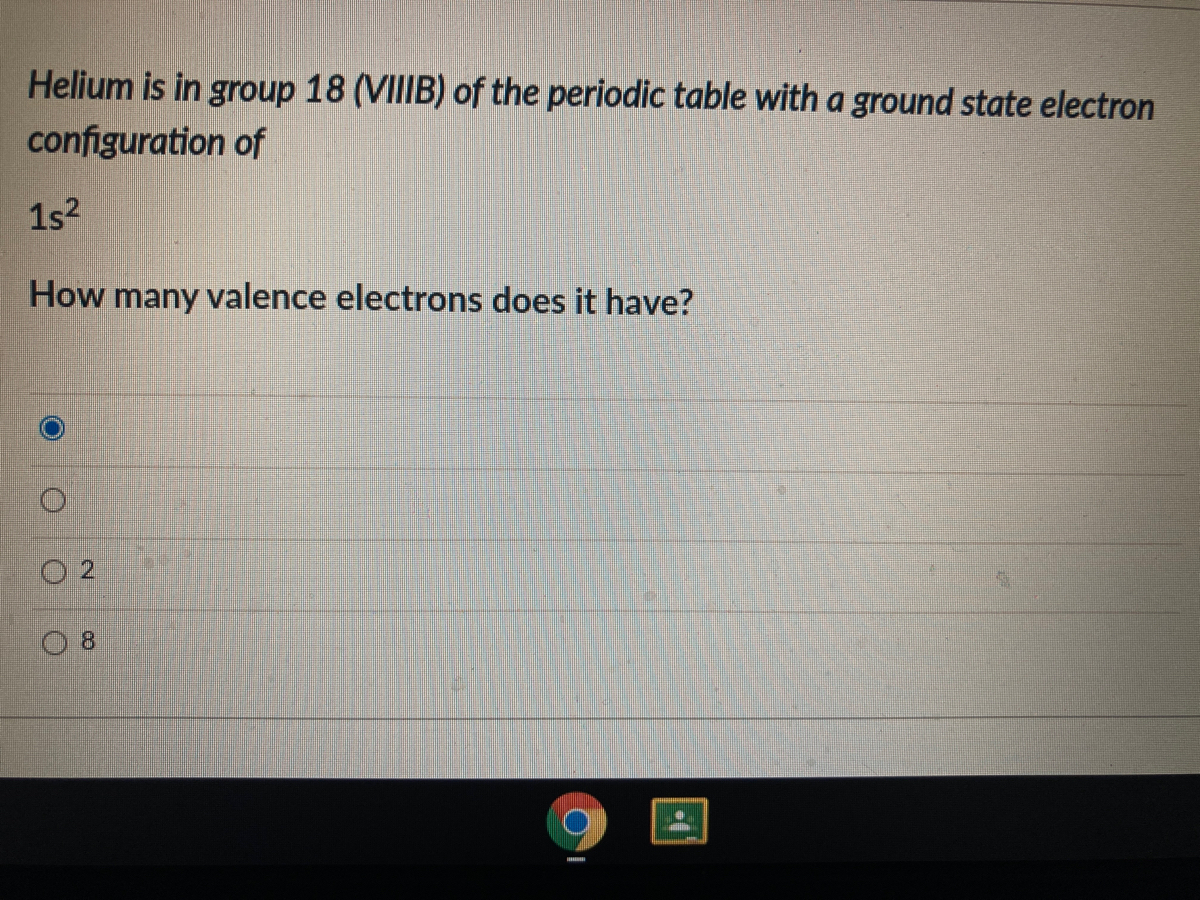
Atomic Number – Protons, Electrons and Neutrons in Helium
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
